|
Browse Chromium(II) chloride and related products at MilliporeSigma. Oxides Dichromate and chromate equilibria is pH dependent: (21.7A.3) HCrO 4 CrO 42 + H + K 10 5.9 H 2 CrO 4 HCrO 4- + H + K10 +0.26 Cr 2 O 72- + H 2 O 2HCrO 4- K10 -2. Reduction of CrX 3 with H 2 /HX gives CrX 2. Before placing in Container D, check the pH with pH-Universal indicator strips (Item No. Synonyms: Chromium dichloride, Chromous chloride, Chromium(II) chloride. Halides CrX 3 are prepared from Cr with X 2, dehydration of CrCl 3. Neutral solutions of the these salts: Container D. P302 + P352: IF ON SKIN: Wash with plenty of water.ĨB Non-combustible, corrosive hazardous materialsġ4 Inorganic salts: Container I. -chloride.jpg)
P301 + P312: IF SWALLOWED: Call a POISON CENTER/doctor if you feel unwell. P261: Avoid breathing dust/ fume/ gas/ mist/ vapours/ spray. 
Unlike other transition metals, the 1+ cation of mercury has the formula Hg22+ and behaves like a polyatomic ion. 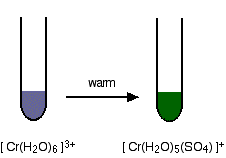
potassium dichromate + hydrochloric acid Potassium chloride + chromium chloride + water +. H411: Toxic to aquatic life with long lasting effects. Learn Test Match Created by Terms in this set (29) What is the formula for mercury (I) chloride The formula for mercury (I) chloride is Hg2Cl2. Write the balanced chemical equation of the following reaction. Chromium (III) Chloride also referred to as chromic chloride describes any of several purple solid compounds with the formula CrCl3 x H2O, where x can be 0, 5, and 6. H317: May cause an allergic skin reaction. Other Name: Chromium chloride (CrCl3),hydrate (1:6) Chromium chloride (CrCl3),hexahydrate Chromic chloride hexahydrate Hexaaquachromium(III) chloride Chromium. Please let us know how we can improve this web app.Chromium(III) chloride anhydrous for synthesis. Related: Molecular weights of amino acids The anhydrous compound with the formula CrCl 3 is a violet solid. Weights of atoms and isotopes are from NIST article. Chromium (III) chloride (also called chromic chloride) describes any of several chemical compounds with the formula CrCl 3 x H 2 O, where x can be 0, 5, and 6.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
